CryoLetters Volume 47 - Issue 3
CryoLetters 47 (3), 162-172 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26310110112
PERSPECTIVE: Frozen and breathless: hypoxia’s role in sperm cryoresistance
Maryna Petrushko1 and Taisiia Yurchuk1,2*
- Institute for Problems of Cryobiology and Cryomedicine of the National Academy of Sciences of Ukraine, Pereyaslavska 23 Str., Kharkiv 61016, Ukraine.
- Institute of Animal Reproduction and Food Research Polish Academy of Sciences in Olsztyn, Trylińskiego 18 Str., Olsztyn 10-683, Poland.
*Corresponding author’s E-mail: taisiya.yur@gmail.com
Abstract
Hypoxia is a critical factor influencing sperm viability by compromising their structural integrity, functional activity, and fertilizing capacity. In reproductive biology and cryobiology, both hypoxia and cryopreservation are known to independently induce cellular stress; however, their combined effects on sperm remain inadequately explored. Here we systematize current knowledge on the effects of hypoxia on sperm cryoresistance, elucidate the molecular mechanisms underlying these effects and evaluate modern strategies for mitigating the combined damage caused by hypoxia and cryopreservation on sperm structure and function. We conducted a comprehensive literature review by analyzing peer-reviewed studies focused on the molecular and cellular responses of mammalian spermatozoa to hypoxia and cryopreservation. Key mechanisms investigated included oxidative stress pathways, mitochondrial function, membrane homeostasis, ion transport and genetic and epigenetic changes. The review also considered current experimental approaches and therapeutic interventions targeting these mechanisms. The analysis revealed that hypoxia disrupts cellular energy metabolism and enhances oxidative stress, leading to reduced sperm survival following cryopreservation. Additionally, cryopreservation itself causes further damage through membrane destabilization, osmotic imbalance and impaired intracellular signaling. These cumulative effects intensify structural and functional deterioration in sperm cells. Emerging mitigation strategies, including antioxidant supplementation, ion regulation, alterations to cryopreservation protocols and the use of hypoxic adaptogens demonstrate potential for improving post-thaw sperm viability. The combined impact of hypoxia and cryopreservation significantly impairs sperm integrity and function, primarily through oxidative and metabolic stress mechanisms. Targeted interventions aimed at counteracting these effects hold promise for enhancing sperm cryoresistance. Finally, this work highlights important avenues for further research and practical applications in reproductive medicine, particularly in fertility preservation and assisted reproductive technologies.
Keywords: cryopreservation; DNA integrity; epigenetic changes; hypoxia; oxidative stress; sperm.
CryoLetters 47 (3), 173-183 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26310110312
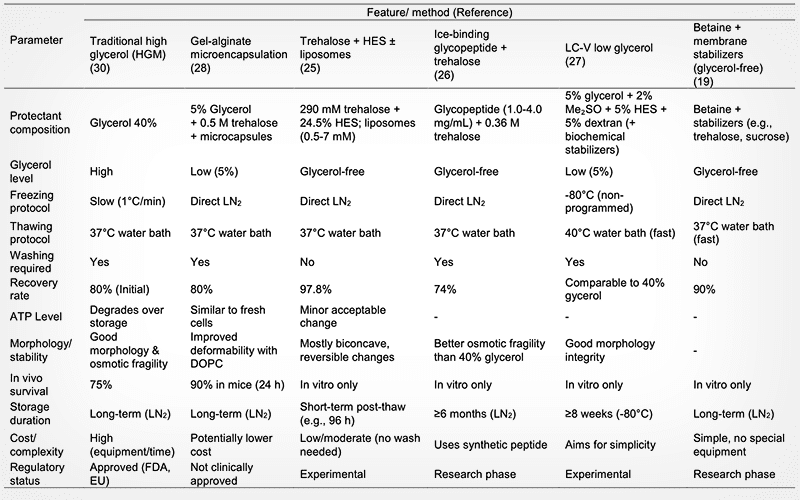
PERSPECTIVE: Advances in glycerol-free and low-glycerol cryopreservation of red blood cells
Ning Wu1, Yijing He1, Weijie Li1,2,3, Wendell Q. Sun1 and Baolin Liu1,2,3*
- Institute of Biothermal Science and Technology, University of Shanghai for Science and Technology.
- Shanghai Technical Service Platform for Cryopreservation of Biological Resources.
- Shanghai Collaborative Innovation Center of Energy Therapy for Tumors, Shanghai 200093, China.
*Corresponding author’s E-mail: blliuk@163.com
Abstract
This review recounts the advances in cryopreservation technologies of red blood cells (RBC). The limitations of traditional glycerol-based methods and the latest developments in cryopreservation with no or less glycerol are elaborated. Although the use of glycerol at high-concentrations effectively prevents ice crystal formation, the associated osmotic stress damages RBC. Moreover, the cumbersome and time-consuming deglycerolization process not only increases operational costs, but also poses the risks of contamination and additonal cell damage, thereby limiting the application of frozen blood for emergency situations. To overcome these challenges, novel strategies have focused on rapid freezing with no or less glycerol, new permeable cryoprotectants and the synergistic use of non-permeable macromolecular/saccharide protectants (such as trehalose and hydroxyethyl starch). These approaches aim to achieve more efficient and safer preservation through multi-faceted protective mechanisms via the reduction of ice crystal damage, osmotic stress and oxidative damage. New strategies demonstrate significant advantages in streamlining processes (eliminating or simplifying deglycerolization steps), improving cell recovery and quality (low hemolysis) and enhancing in vivo efficacy. However, challenges such as long-term storage stability, scale-up production costs, standardization and regulatory approval remain critical issues to be addressed before clinical translation can be realized. Future research needs to focus on optimizing cryoprotectant formulations, elucidating molecular mechanisms, establishing a standardized quality control system and clinical validation, thereby revolutionizing blood inventory management.
Keywords: cryopreservation; cryoprotectants; glycerol-free CPAs; low-glycerol CPAs; red blood cells.
CryoLetters 47 (3), 184-200 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26310110612
Synergistic effect of L-carnitine, sodium pyruvate and casein supplementation in Tris-based extender on freezeability of Barbari buck semen
Shivika Chouksey1, Satya Nidhi Shukla2*, Sanju Mandal3, Amita Dubey4 and Ayushi Chourasia5
- Department of Veterinary Gynaecology & Obstetrics, College of Veterinary Science & Animal Husbandry, NDVSU, Jabalpur (M.P.), 482002, India.
- M.P. State Livestock & Poultry Development Corporation, Bhopal, M.P., India & Department of Veterinary Gynaecology & Obstetrics, CVSc & AH., NDVSU, Jabalpur, 482002, India.
- Department of Veterinary Physiology & Biochemistry, College of Veterinary Science & Animal Husbandry, NDVSU, Jabalpur (M.P.), 482002, India.
- Department of Veterinary Pathology, College of Veterinary Science & Animal Husbandry, NDVSU, Jabalpur (M.P.), 482002, India.
- Department of Veterinary Pharmacology & Toxicology, College of Veterinary Science & Animal Husbandry, NDVSU, Jabalpur (M.P.), 482002, India.
*Corresponding authors’ E-mail: snshukla2@gmail.com
Abstract
Background
Post-thaw quality of frozen buck sperm is strongly influenced by the choice of diluent and antioxidant supplementation. L-carnitine, sodium pyruvate and casein enhance sperm cryopreservation by providing antioxidant protection, stabilizing membranes, supporting mitochondrial energy metabolism and preserving DNA integrity during the freeze–thaw process.
Objective
This study aimed to assess the combined effect of L-carnitine, casein and sodium pyruvate addition in semen extenders on cryopreserved buck semen survivability, in vitro sperm parameters and oxidative stress at various months and seasons in a tropical area.
Materials and methods
A total of 12 adult Barbari bucks were used in this study. Semen was cryopreserved using an extender supplemented with L-carnitine (2 mM), sodium pyruvate (1 mM), and casein (0.068 g). Post-thaw semen was evaluated for volume, sperm concentration, mass motility, progressive motility, viability, plasma membrane integrity (HOST), DNA integrity, capacitation and apoptosis status, mitochondrial membrane potential (MMP), lipid peroxidation (LPO), and superoxide dismutase (SOD) activity.
Results
Supplementation significantly improved post-thaw sperm motility, viability, plasma and acrosomal membrane integrity, mitochondrial function, and DNA stability while reducing apoptosis and cryocapacitation compared to the control group. Seasonal variations were observed, with semen collected during September–November and the rainy season showing superior parameters, followed by the transition from rainy to winter. The antioxidants effectively mitigated oxidative stress and preserved mitochondrial and DNA integrity throughout the freeze–thaw process.
Conclusion
The inclusion of L-carnitine, sodium pyruvate, and casein in extenders significantly improved post-thaw buck sperm quality by preserving motility, membrane integrity, mitochondrial function, and DNA stability while reducing oxidative stress and apoptotic-like changes. These findings highlight the potential of this formulation to enhance the fertilizing capacity of cryopreserved semen across seasonal variations.
Keywords: buck semen; L-carnitine; casein; cryopreservation.
CryoLetters 47 (3), 201-213 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26310110712
Histological and immunohistochemical effects of dynamic plate cryolipolysis on localized abdominal adiposity: a case study with multimodal cellular analysis
Christiane Rodrigues Tofoli Palauro1, Patrícia Froes Meyer2*, Rafaella Rêgo Maia3, Eneida de Morais Carreiro2, Ciro Dantas Soares2, Flávio de Paiva Dumaresq4, Fernando Cesar Câmara de Oliveira3 and Fernanda Cabegi de Barros5
- Vila Velha University, Vila Velha, Brazil.
- International Research Group, Natal, Brazil.
- Federal University of Rio Grande do Norte, Natal, Brazil.
- State University of Health Sciences of Alagoas, Maceió, Brazil.
- Inspirar College, São Paulo, Brazil.
*Corresponding author’s E-mail: patricia.froesmeyer@gmail.com
Abstract
Background
Dynamic plate cryolipolysis (DPC) has emerged as a non-invasive alternative technique for treating localized adiposity, particularly in anatomically challenging areas.
Objective
To evaluate the effects of DPC on the cutaneous and subcutaneous tissues of the abdominal region through histological and immunohistochemical analyses.
Materials and methods
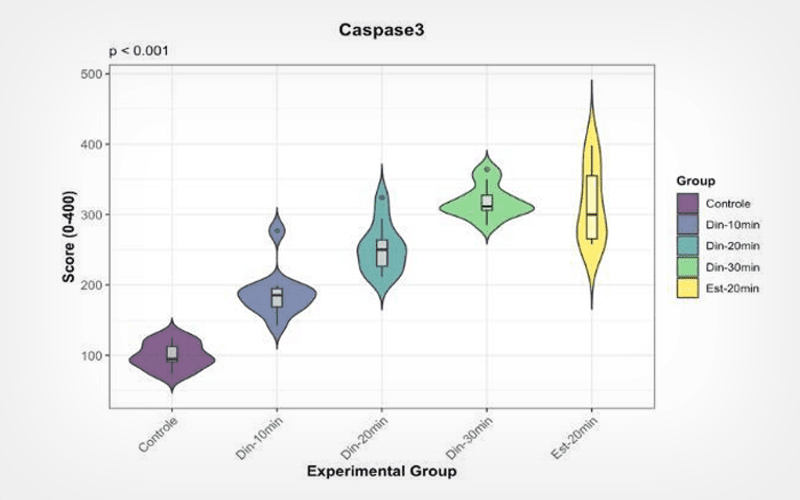
Four volunteers underwent distinct protocols of DPC (10, 20 or 30 min) or static cryolipolysis (20 min). DPC protocols were performed at -5°C, followed by 3 min of reperfusion and manual massage. The right infraumbilical region was treated, while the contralateral side served as control. Samples were collected during elective abdominoplasty and analyzed for biomarkers of apoptosis, inflammation, fibroblast activity, collagen remodeling, heat stress response, and metabolic or hormonal modulation.
Results
DPC induced adipocyte apoptosis, confirmed by detection of caspase-3, COX-2, and macrophage (CD68 and CD163) expression, fibroblast activation (FGF2 and FGFR1), and a predominance of type I collagen deposition. Additionally, modulation of metabolic and hormonal markers was observed, including reduced PPAR-γ and aromatase expression, and partial UCP1 expression, suggesting metabolic modulation with potential thermogenic effects.
Conclusion
DPC induced significant and controlled cellular responses, supporting its safety and therapeutic potential. Beyond aesthetic applications, these findings point to broader clinical implications. Larger-scale studies are required to validate and expand these findings.
Keywords: apoptosis; dynamic cryolipolysis; immunohistochemistry; localized adiposity; tissue remodeling.
CryoLetters 47 (3), 214-223 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26310110212
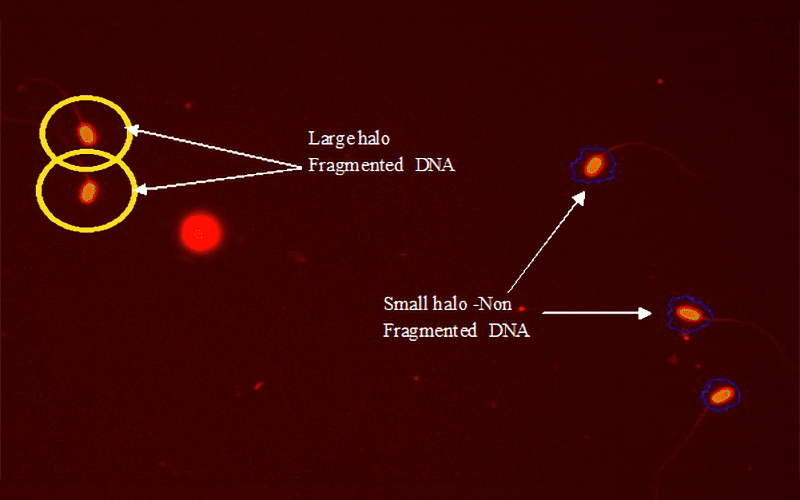
Cryotolerance of buck goat spermatozoa: functional and oxidative biomarkers across different age groups
Yashpal Chaudhary1, Mukul Anand1*, Sarvajeet Yadav1, Shalini Vaswani2, Rahul Dhariya1, Brijesh Yadav1, Amit Kumar1, Rashmi Singh1, Shikha Pareek1 and Anchal Sharma1
- Department of Veterinary, Physiology College of Veterinary Sciences and Animal Husbandry, Pandit Deen Dayal Upadhayaya Pashu Chikitsa Vigyan Vishwavidyalaya Evam Go Anusandhan Sansthan, Mathura, U.P., India.
- Department of Animal Nutrition, College of Veterinary Sciences and Animal Husbandry, Pandit Deen Dayal Upadhayaya Pashu Chikitsa Vigyan Vishwavidyalaya Evam Go Anusandhan Sansthan, Mathura, U.P., India.
*Corresponding author’s E-mail: drmukulanandvet@gmail.com
Abstract
Background
Cryopreservation is integral to artificial insemination and germplasm conservation in goats, yet post-thaw sperm survival remains inconsistent due to structural and oxidative damage. Age, a key intrinsic factor, critically modulates sperm functional competence and antioxidative capacity, making it a potential predictor of cryotolerance. Understanding age-associated functional and oxidative biomarkers can refine buck selection strategies to enhance fertility outcomes.
Objective
To systematically evaluate the influence of age on semen quality, oxidative stress markers, and functional competence of spermatozoa in both fresh and cryopreserved semen of Barbari bucks, with the aim of identifying age-associated functional and oxidative biomarkers predictive of sperm cryotolerance and post-thaw survival.
Materials and methods
Eighteen breeding bucks were categorized into three groups: Group I (15 to 25-month young adults), Group II (26 to 40-month mid adults), and Group III (41- to 55-month old adults). Fresh and post-thaw semen was analyzed using CASA, flow cytometry, and biochemical assays for motility, viability, acrosomal and plasma membrane integrity, mitochondrial activity, DNA fragmentation, capacitation status, and antioxidative enzyme activity.
Results
Mid-aged bucks (26–40 months) showed significantly higher acrosomal integrity, antioxidant enzyme activity and genomic stability with better cryotolerance compared with younger and older bucks. Younger bucks exhibited incomplete functional maturation, while older bucks displayed oxidative deterioration, higher DNA fragmentation, and reduced antioxidant defense.
Conclusion
The optimal age window for semen collection in bucks is mid-aged bucks (26 to 40-month old). Functional and oxidative biomarkers provide better predictors of cryotolerance than conventional semen traits, offering actionable insights for AI programs and germplasm conservation.
Keywords: age effect; bucks; oxidative stress; sperm cryopreservation.
CryoLetters 47 (3), 224-234 (2026)
© CryoLetters, editor@cryoletters.org
doi.org/10.54680/fr26310110412
Me2SO-free multicomponent cryoprotectants for efficient cryopreservation of Jurkat cells
Zixuan Zhang1,2,3, Qiang Xu1,2,3 and Xinli Zhou1,2,3*
- Institute of Biothermal Science & Technology, University of Shanghai for Science and Technology, Shanghai 200093, China.
- Shanghai Co-innovation Center for Energy Therapy of Tumors, Shanghai 200093, China.
- Shanghai Technical Service Platform for Cryopreservation of Biological Resources, Shanghai 200093, China.
*Corresponding author’s E-mail: zjulily@163.com
Abstract
Background
Jurkat cells, a human T lymphocyte line widely used in immunological studies, require reliable cryostorage to maintain post-thaw quality. Dimethyl sulfoxide (Me2SO) is the standard cryoprotectant, but its cytotoxicity may impair cell performance after thawing.
Objective
To develop a Me2SO-free multicomponent cryoprotectant agent (CPA) for Jurkat cell cryopreservation.
Materials and methods
A Me2SO-free CPA composed of ethylene glycol (EG), disaccharides (trehalose, sucrose, glucose), and amino acids (histidine, isoleucine, tetrahydropyrimidine) was formulated. The effects of CPA composition and concentration on Jurkat cell cryopreservation were evaluated. Post-thaw viability and proliferation capacity were measured to identify optimal formulations. Cryomicroscopy was used to observe ice crystal growth and recrystallization under different CPA conditions.
Results
Disaccharides at 0.1 M improved post-thaw viability, whereas higher concentrations caused a marked decline in viability, indicating concentration-dependent cryoinjury. Among the disaccharides tested, trehalose showed the most consistent protection. Adding 20 mM histidine to 12.5% EG + 0.1 M trehalose increased viability from 87.8% to 93.8% (p < 0.05). Reducing EG to 10% while maintaining trehalose and histidine preserved viability above 91%, comparable to 10% Me2SO. Proliferation assays confirmed sustained recovery, with 40 mM histidine producing the highest proliferation. Cryomicroscopy showed that trehalose and histidine suppressed ice crystal growth and recrystallization.
Conclusion
The optimized Me2SO-free, EG-based multicomponent CPA provides effective cryoprotection for Jurkat cells with reduced cytotoxicity and represents a practical alternative to Me2SO-based protocols.
Keywords: cryopreservation; ice crystallization inhibition; Jurkat cells; Me2SO-free cryoprotectant.






